The Head of Oncology and her Clinical Operations team at the Sponsor company were facing increasing competition for patients in their oncology trials. The complexity of the protocols and the length of the trials made it difficult to track patients longitudinally prior to randomization, leading to major blind spots in the patient recruitment and enrollment funnel.
The Sponsor also assumed that their sites were working on their studies, but lacked the data to know which sites were successfully recruiting and pre-screening and which were not. They found that few candidates were actually hitting the IxRS system.
The Sponsor deployed StudyTeam globally to their research sites across three key oncology trials
With the help of StudyTeam’s Customer Success team, each site was contacted to activate and train the coordinators on StudyTeam’s simple user interface; sites were up and running on StudyTeam within just a few weeks.
The sites leveraged the platform’s tagging and follow-up features to create follow up tasks for each patient, be alerted when they came due, and ensure that they didn’t lose track of patients who become eligible after the longer tracking periods that are typical in oncology studies. Sites were also able to use StudyTeam as a searchable patient database to quickly and easily screen patients across multiple trials; those patients who had screened out of one study could be instantly considered for another.
Because uptake of StudyTeam was nearly universal across hundreds of the Sponsor’s sites, the Sponsor could now see which sites were having success with recruitment and which were struggling, allowing them to focus their site management efforts on sites needing assistance.
StudyTeam’s Trial Board made it easy for the Sponsor to track oncology patients over long periods of time and during lengthy medication washout periods. This ensured that they were able to enroll every enrollable patient.
Because sites chose StudyTeam overwhelmingly over other methods for tracking recruitment, prescreening, and enrollment, the Sponsor had access to comprehensive real-time enrollment funnel data.
StudyTeam’s Visit Window Calculator made it easy for sites to schedule patients within visit tolerances. This saved sites immense amounts of time and reduced protocol deviations for the Sponsor.
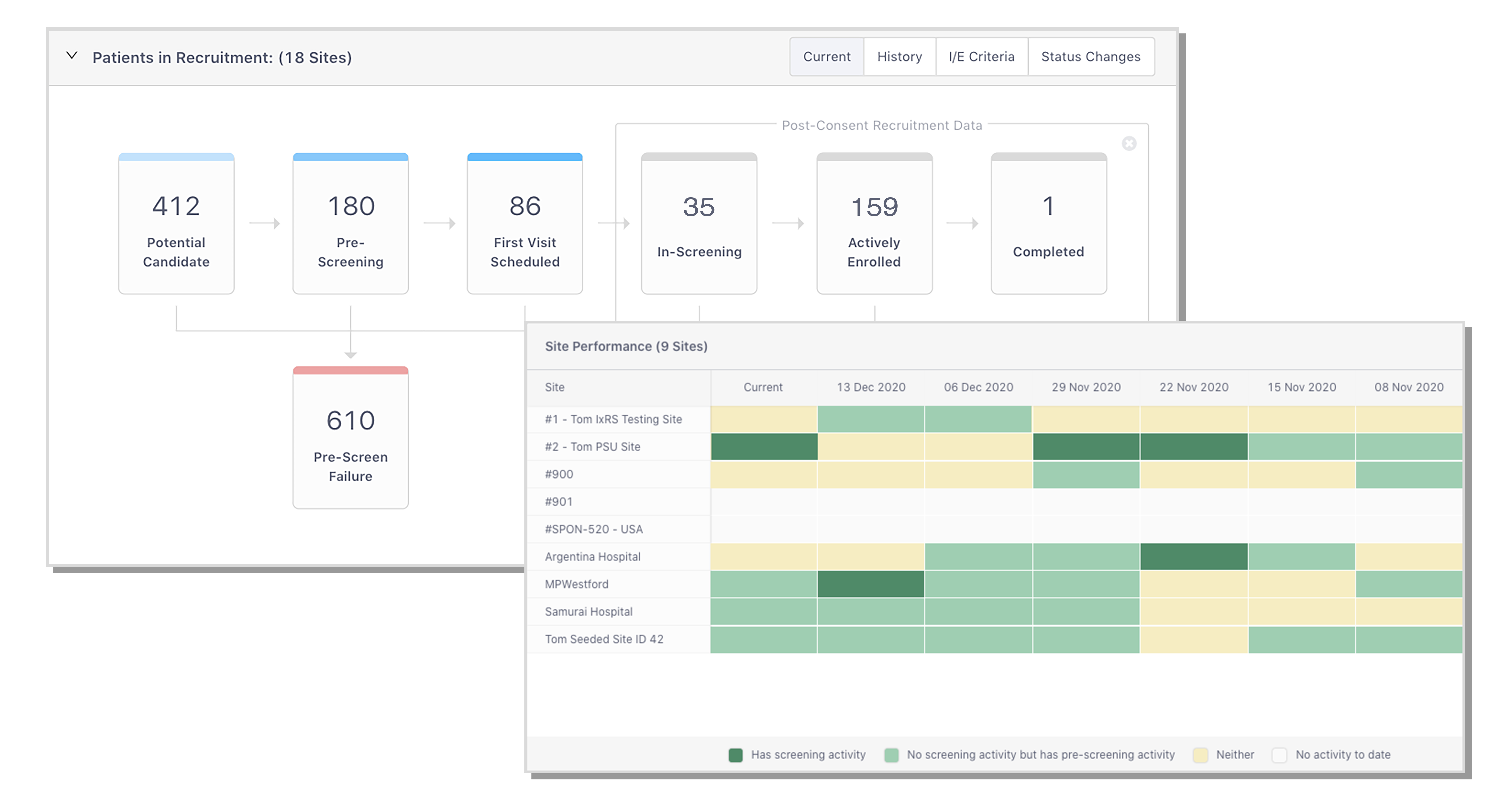
A sample Trial Dashboard illustrating the types of real-time data sponsors receive in StudyTeam.
StudyTeam’s Trial Board provides sites with a central place where all identified patient candidates stay visible until they are eligible. Making it easier to track patients over long medication washout periods. Helping you enroll every enrollable patient.
StudyTeam’s Visit Window Calculator makes it easy for sites to schedule patients within visit tolerances, even when changes occur. Saves massive amounts of time for sites. Reduces protocol deviations for sponsors.
StudyTeam makes it easy for sites to pre-screen patients for multiple trials. Helping sites find the best course of treatment for their patient, and ensure sponsors enroll every enrollable patient.
The Site Heat map and Patient Recruitment Source report ensure you know, earlier, which sites are having success with recruitment and which are not, allowing you to develop focused outreach strategies.
Gain insights and control that you’ve never had before. A clinical trial enrollment software that both sponsors and sites will love.
.png?width=65&name=OST%20Transparent%20(1).png)
OneStudyTeam, a member of the Reify Health portfolio, provides the cloud-based platform StudyTeam to accelerate the development of new and life-saving therapies. StudyTeam brings research site workflows online and enables sites, sponsors, and other key stakeholders to work together more effectively using common technology. The suite of StudyTeam solutions reduces site burden and helps sites pre-screen and enroll more patients, provides sponsors with end-to-end visibility into recruitment activity across all channels, and guides sites in conducting the trial for patients who have been enrolled. StudyTeam is trusted by the largest global biopharmaceutical companies, used in more than 6,000 research sites, and is available in over 100 countries.
One mission. One team. That’s OneStudyTeam.
33 Arch Street 17th Floor Boston, MA 02110
Join Our Mailing List
Get OneStudyTeam news and clinical trial enrollment insights delivered directly to your inbox.
Copyright © 2024 OneStudyTeam, Inc. All Rights Reserved.